Accurate volume measurement is critical in acid-base analysis, enabling precise quantification of solutions. Titrations rely on knowing volumes of acid and base for concentration determination.
Volumetric glassware, like burettes and pipettes, are essential for precise acid volume delivery and sample transfer during chemical analysis procedures.
Importance of Accurate Volume Measurement
Precise volume measurement is paramount in acid-base titrations, directly impacting the reliability of results. Errors in volume readings propagate through calculations, leading to inaccurate concentration determinations. Titrations, as highlighted by Chemistry LibreTexts, depend on knowing the volumes of both acid and base to calculate the unknown concentration.
Volumetric glassware, including burettes and pipettes, are designed for accuracy, but proper technique is crucial. Parallax errors, for instance, can significantly skew readings. Furthermore, understanding the equivalence point – where acid and base neutralize – relies on careful volume tracking. Accurate measurements ensure the endpoint closely reflects the equivalence point, maximizing the precision of the analysis.
Ultimately, meticulous volume measurement is not merely a procedural step, but a cornerstone of quantitative chemical analysis.
Applications in Chemical Analysis
Acid-base titrations, a core technique in chemical analysis, heavily rely on accurate acid volume measurement. These titrations are used to determine the concentration of unknown acid or base solutions, as explained by Chemistry LibreTexts, by comparing them to a standard solution.
Applications span diverse fields, including environmental monitoring – assessing acidity in water samples – and pharmaceutical quality control – ensuring drug purity and concentration. Volumetric analysis, employing titration, is a quantitative method crucial for research and industrial processes.
Beyond simple concentration determination, titrations can reveal the purity of chemical compounds and identify unknown substances. Precise volume measurements, facilitated by burettes and pipettes, are therefore fundamental to obtaining reliable analytical data across numerous scientific disciplines;

Core Principles of Acid-Base Titration
Titration involves reacting an acid with a base to reach the equivalence point, signaled by an indicator’s color change, determining an unknown concentration.
Standard solutions of known concentration are vital, alongside precise volume measurements, for accurate calculations in these quantitative analyses.
Understanding Equivalence Point and End Point
The equivalence point in a titration represents the complete stoichiometric reaction between the acid and base, where moles of acid equal moles of base. However, directly observing this point is often difficult. This is where the end point comes into play – it’s the observable change, typically a color shift from an indicator, signaling the titration’s completion.
Ideally, the end point closely approximates the equivalence point, but discrepancies can occur due to indicator limitations. Accurate volume measurements, using burettes, are crucial for pinpointing the end point and, consequently, estimating the equivalence point. Knowing volumes of both acid and base, alongside the standard solution’s concentration, allows for precise concentration determination of the unknown solution. The indicator signals neutralization (H+ = OH–) via color change.
Therefore, careful observation and precise volume control are paramount for reliable titration results.
Role of Standard Solutions
Standard solutions are the cornerstone of accurate acid-base titrations. These solutions have precisely known concentrations, meticulously determined through calibration against primary standards. They serve as the benchmark against which unknown solutions are compared, enabling quantitative analysis. Volumetric analysis, or titrimetry, fundamentally relies on comparing an unknown solution with a standard.
The concentration of the standard solution is critical; any error here directly propagates to the final calculation of the unknown’s concentration. Precise volume measurements, achieved with burettes and pipettes, are essential when using standard solutions. Knowing the exact volume of standard solution reacting with the analyte allows for accurate determination of the analyte’s concentration.
Ultimately, the reliability of a titration hinges on the accuracy of the standard solution and the precision of volume measurements.
pH and pOH Relationship in Titrations
During titrations, the pH of the solution changes as acid and base react. Understanding the relationship between pH and pOH is crucial for interpreting these changes and accurately determining the equivalence point. The fundamental equation pH + pOH = 14.00 allows for easy conversion between these values, providing a complete picture of the solution’s acidity or basicity.
As titration progresses, monitoring pH changes reveals the neutralization process. Knowing the pH allows calculation of pOH, and vice versa, offering redundant data for verification. This relationship is particularly useful when dealing with strong acids and bases, where pH changes are more dramatic.
Furthermore, understanding the dissociation constants, KA and KB, and their relationship to pKA and pKB, aids in analyzing the titration curve and identifying the equivalence point accurately.
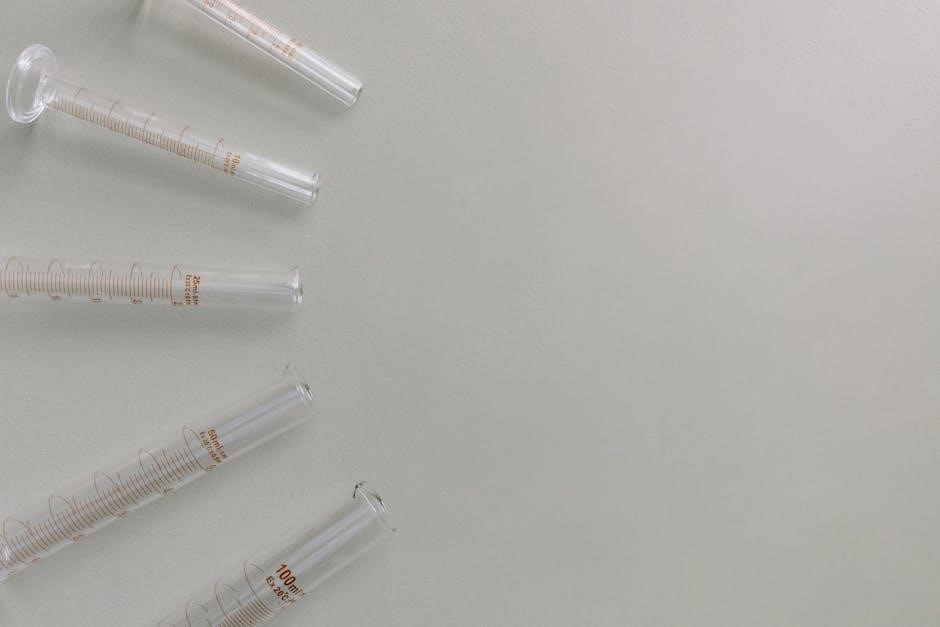
Essential Equipment for Acid Volume Measurement
Precise acid volume measurement requires specialized glassware: burettes for delivery, pipettes for transfer, and volumetric flasks for preparation of standard solutions.
Beakers and Erlenmeyer flasks serve as reaction vessels, facilitating controlled mixing during titration processes for accurate analysis.
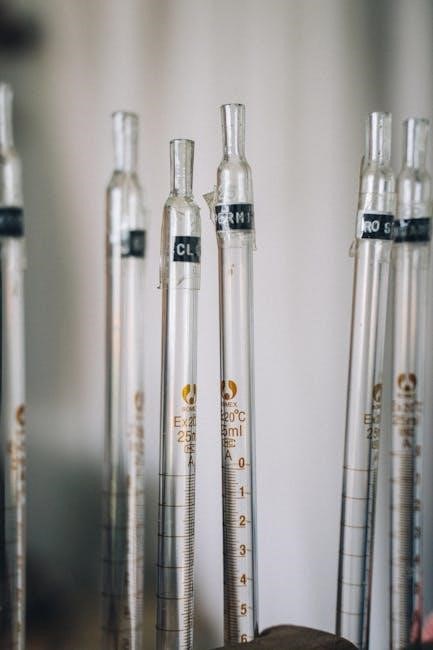
Burettes: Precision Volume Delivery
Burettes are graduated glass tubes, featuring a stopcock for precise and controlled dispensing of variable volumes of liquid. They are essential for accurate titrations, allowing for the gradual addition of a standard solution – often an acid – to an analyte until the reaction’s endpoint is reached.
Calibration is crucial; burettes must be accurately calibrated to ensure volume readings are reliable. Reading the meniscus correctly, avoiding parallax error, is also vital for precision. Burettes are typically used in conjunction with a stand to maintain stability during the titration process. Their design allows for highly accurate volume delivery, making them indispensable in quantitative chemical analysis.
Different sizes are available, catering to varying titration volumes, and are constructed from glass or plastic, depending on the application and chemical compatibility.
Pipettes: Accurate Sample Transfer
Pipettes are designed for the precise transfer of a fixed or variable volume of liquid. Unlike burettes, which deliver liquid gradually, pipettes transfer a predetermined volume in a single operation. They are critical for accurately measuring the analyte solution during titration, ensuring the correct stoichiometric ratio for the reaction.
Several types exist, including volumetric (fixed volume), graduated (variable volume), and micropipettes for very small volumes. Accuracy depends on proper technique – ensuring complete aspiration and expulsion of the liquid, and avoiding air bubbles. Like burettes, pipettes require regular calibration to maintain their accuracy.
Proper cleaning and maintenance are also essential to prevent contamination and ensure reliable results in acid-base titrations and other quantitative analyses.
Volumetric Flasks: Precise Solution Preparation
Volumetric flasks are specifically designed to prepare solutions of known concentration with high accuracy. These flasks have a long neck and a bulbous body, featuring a single calibration mark indicating the precise volume the flask contains when filled to that line. They are essential for creating standard solutions used in titrations, where knowing the exact concentration of one solution is paramount.
To use a volumetric flask correctly, dissolve the solute in a portion of solvent, then add solvent until the liquid level approaches the calibration mark. Finally, carefully add solvent dropwise until the meniscus aligns with the mark, ensuring eye-level observation to avoid parallax error.
Thorough mixing is crucial after reaching the calibration mark to ensure homogeneity and accurate concentration.
Beakers and Erlenmeyer Flasks: Reaction Vessels
Beakers and Erlenmeyer flasks, while not designed for precise volume measurement, serve as crucial reaction vessels during acid-base titrations and solution preparation. Beakers, with their wide openings, are ideal for mixing and heating liquids, though they are unsuitable for accurate volume containment due to their broad shape.
Erlenmeyer flasks, characterized by their conical shape, are preferred for titrations. This design minimizes liquid loss during swirling, ensuring thorough mixing of reactants. The narrow neck also reduces the risk of spills and allows for easier addition of reagents.
Although not for precise measurement, graduated markings on some flasks offer approximate volume estimations for convenience during experiments.

Indicators in Acid-Base Titrations
Indicators, like phenolphthalein, signal neutralization via color change at the equivalence point. Accurate endpoint determination relies on selecting the appropriate indicator for the titration.
Phenolphthalein Indicator: Properties and Use
Phenolphthalein is a widely used indicator in acid-base titrations, notably with dilute hydrochloric acid and sodium hydroxide solutions. It’s colorless in acidic solutions (pH < 8.3) and transitions to a vibrant pink-magenta color in basic solutions (pH > 8.3).
This distinct color change allows for clear endpoint detection during titration. The indicator functions by changing its molecular structure based on the solution’s pH, resulting in differing light absorption properties.
Care must be taken as the color change can be subtle, and factors like indicator concentration and lighting conditions can influence visibility. Proper use involves adding a few drops to the analyte solution before titration begins, ensuring the endpoint is accurately observed.
Choosing the Right Indicator for the Titration
Selecting the appropriate indicator is crucial for accurate titration results. The indicator’s pH range must overlap with the steep portion of the titration curve, near the equivalence point. Phenolphthalein is suitable for strong acid-strong base titrations, but other indicators are better for weak acid/base combinations.
Consider the expected pH at the equivalence point. Methyl orange, for example, changes color in a more acidic range than phenolphthalein. Carefully review the indicator’s color change range and ensure it aligns with the titration’s chemistry.
Factors like solution color and desired precision also influence indicator choice. A clear color change is essential for accurate endpoint determination, minimizing subjective error during observation.
Color Change and Endpoint Determination
Endpoint determination relies on observing the indicator’s distinct color change, signaling neutralization. This isn’t precisely the equivalence point, but a close approximation. Careful observation is key; the first persistent color change indicates the endpoint.
The color change should be clearly visible against the solution background. Stirring ensures thorough mixing and a uniform color. Titration is stopped immediately upon observing the endpoint to avoid overshooting.
Accuracy improves with dropwise addition of titrant near the expected endpoint. Multiple titrations are recommended, calculating the average volume used for a more reliable result. Record observations meticulously for data analysis and error assessment.

Titration Techniques and Procedures
Dilute hydrochloric acid and sodium hydroxide solutions are commonly titrated using phenolphthalein as an indicator, following a precise, step-by-step process.
Titration involves careful addition of a titrant to an analyte until the reaction reaches completion, indicated by a color change.
Dilute Hydrochloric Acid Titration
Dilute hydrochloric acid (HCl) titration is a common acid-base neutralization procedure. It typically involves titrating the HCl solution with a standardized base, such as sodium hydroxide (NaOH). Precise volume measurements are paramount, utilizing a burette for controlled addition of the base.
Phenolphthalein serves as a visual indicator, transitioning from colorless in acidic conditions to pink at the endpoint, signifying neutralization (when H+ = OH–). The equivalence point, where moles of acid equal moles of base, is determined by observing this color change.
Accurate readings from the burette, avoiding parallax error, are crucial for calculating the HCl concentration. Knowing the volumes and the base’s concentration allows for determining the unknown acid’s concentration through stoichiometric calculations.
Dilute Sodium Hydroxide Solution Titration
Titrating dilute sodium hydroxide (NaOH) solutions involves reacting them with a standardized acid, commonly dilute hydrochloric acid (HCl). This neutralization reaction is carefully monitored to determine the NaOH concentration. Precise volume delivery is achieved using a burette to add the HCl solution incrementally.
Phenolphthalein indicator is frequently employed, exhibiting a color change from pink in basic solutions to colorless upon reaching the endpoint, indicating complete neutralization. Accurate endpoint detection is vital for reliable results.
Careful burette readings, minimizing parallax error, and knowing the HCl concentration allow for stoichiometric calculations to determine the NaOH concentration. Volumetric glassware calibration ensures accuracy, while temperature considerations impact solution volumes.
Step-by-Step Titration Process
Begin by accurately pipetting a known volume of the unknown acid or base solution into a flask. Add a few drops of a suitable indicator, like phenolphthalein, to visualize the endpoint. Fill a burette with a standardized solution of the opposing reactant – acid for base titration, or base for acid titration.
Slowly add the titrant from the burette, swirling the flask continuously, until the indicator undergoes a persistent color change, signaling the endpoint. Record the volume of titrant used with precision. Repeat the titration multiple times for improved accuracy and reliability.
Calculate the concentration of the unknown solution using stoichiometric principles and the recorded titration data. Ensure proper handling and disposal of chemicals, adhering to safety protocols throughout the process.

Volume Measurement Errors and Mitigation
Parallax errors during burette readings and temperature fluctuations impacting volume are common issues. Calibration of glassware and careful reading techniques minimize these inaccuracies.
Accurate volume measurements are crucial; therefore, addressing these errors ensures reliable titration results and precise concentration determinations.
Parallax Error in Burette Readings
Parallax error occurs when the observer’s eye isn’t directly level with the liquid meniscus in a burette. This leads to an inaccurate volume reading, either overestimating or underestimating the actual volume dispensed. Proper technique dictates positioning the eye at the same height as the meniscus to obtain a true reading.
To mitigate parallax, burettes often feature markings or a dark background to enhance meniscus visibility. Consistent eye level is paramount during measurements. Furthermore, using a white card behind the burette can improve contrast and reduce reading errors. Ignoring this error can significantly impact titration accuracy, leading to incorrect concentration calculations. Regular practice and awareness of this phenomenon are essential for precise acid-base titrations.
Ultimately, minimizing parallax ensures reliable and reproducible results in quantitative chemical analysis.
Calibration of Volumetric Glassware
Volumetric glassware, including burettes, pipettes, and flasks, requires periodic calibration to ensure accuracy. Calibration involves comparing the actual volume delivered or contained by the glassware to its nominal (stated) volume. This is typically done using a calibrated balance and distilled water, determining the mass of water equivalent to a known volume.
Deviations from the nominal volume are recorded and can be applied as corrections during subsequent measurements. Factors like temperature fluctuations and glassware aging can affect volume accuracy, necessitating regular calibration. Traceability to national standards is crucial for reliable results. Proper calibration minimizes systematic errors and enhances the precision of acid-base titrations.
Maintaining calibration records is essential for quality control and data integrity in analytical chemistry.
Temperature Effects on Volume
Temperature significantly impacts the volume of liquids, including acids and bases used in titrations. Most liquids expand when heated and contract when cooled, altering their density and therefore, their volume. Volumetric glassware is calibrated at a specific temperature, typically 20°C.
Measurements made at temperatures differing from the calibration temperature require corrections to account for thermal expansion or contraction. These corrections are particularly important for high-precision work. Temperature variations can introduce systematic errors if not addressed.
Maintaining a consistent temperature during titrations, or applying appropriate temperature correction factors, is crucial for accurate volume measurements and reliable analytical results.

Alternative Volume Measurement Methods
Cell volume can be determined using dye displacement techniques, employing dyes like Acid Blue 9. Transmission image analysis quantifies volume changes via dye contrast.
These methods offer alternatives to traditional volumetric glassware for specific applications, particularly in biological studies.
Cell Volume Measurement using Dye Displacement
Dye displacement offers a unique approach to determining cell volume, differing from traditional acid-base titration methods. This technique involves suspending cells in a strongly colored dye solution, notably Acid Blue 9, which is favored for its low cost and minimal impact on cell viability at working concentrations.
Intact cells displace the dye, creating a lighter-colored region proportional to their volume. Transmission image analysis is then employed, capturing images at a wavelength maximizing dye absorption. By analyzing the change in light transmission, researchers can accurately calculate the volume of individual cells. This method is particularly useful when dealing with biological samples where precise acid-base titration isn’t feasible or desirable.
Equipment includes a microscope with imaging capabilities, a light source, and software for image analysis.
Acid Blue 9 Dye for Cell Volume Analysis
Acid Blue 9 is a frequently utilized dye in cell volume measurements via displacement, offering several advantages for biological applications. Its primary appeal lies in its affordability and, crucially, its demonstrated lack of significant cytotoxic effects on mammalian cells at the concentrations needed to generate sufficient contrast for accurate readings – typically around 0.7.
The dye’s strong coloration allows for clear visualization of the displacement created by cells, enabling precise volume determination through transmission image analysis. Compared to other dyes, Acid Blue 9 provides a favorable balance between visibility and biocompatibility, making it a preferred choice for researchers.
Its properties facilitate reliable and non-invasive cell volume assessment, complementing traditional acid-base titration techniques.
Transmission Image Analysis in Dye Displacement
Transmission image analysis plays a vital role in quantifying cell volume when employing dye displacement methods, particularly with Acid Blue 9. This technique involves capturing images of the dye solution after cells have been introduced, utilizing a wavelength corresponding to the dye’s maximum absorption;
By analyzing the changes in light transmission caused by the displaced dye, researchers can accurately calculate the volume occupied by individual cells. Sophisticated image processing software is often used to delineate cell boundaries and quantify the area of dye displacement.
This method provides a non-invasive and relatively simple approach to cell volume measurement, complementing traditional acid-base titration methods and offering valuable insights into cellular physiology.
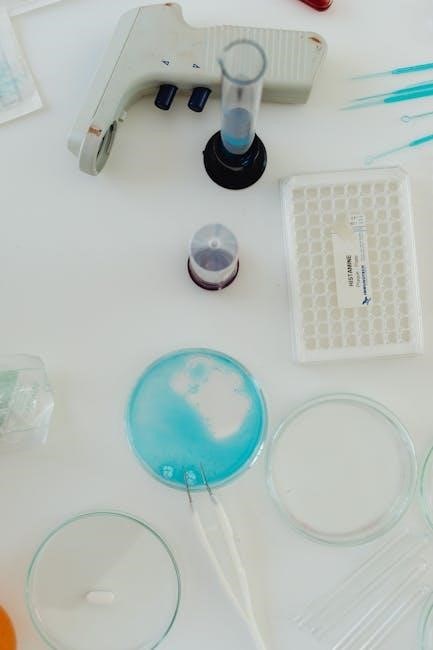
Data Analysis and Calculations
Titration data allows for precise concentration determination of unknown solutions. Calculations involve converting pH to pOH and utilizing KA/KB values for accurate results.
Knowing volumes of acid and base, alongside standard solution concentrations, is fundamental for quantitative analysis in volumetric chemical analysis.
Determining Concentration from Titration Data
Accurately determining the concentration of an unknown acid or base solution hinges on meticulous titration data analysis. The core principle involves utilizing the volumes of both the titrant (standard solution) and the analyte (unknown solution) at the equivalence point. This point, signaled by an indicator’s color change, signifies complete neutralization.
Calculations employ the stoichiometry of the reaction. Knowing the molarity and volume of the standard solution allows for calculating the moles of titrant used; From this, and the balanced chemical equation, the moles of analyte can be determined. Finally, dividing the moles of analyte by its volume yields the unknown concentration.
Precise volume measurements, achieved with burettes and pipettes, are paramount for reliable results. Any errors in volume readings directly impact the calculated concentration, emphasizing the importance of careful technique and calibrated glassware.
Converting pH to pOH Values
Understanding the relationship between pH and pOH is fundamental in acid-base chemistry, particularly during titration analysis. These values represent the acidity and basicity of a solution, respectively, and are intrinsically linked through the ion product of water (Kw).
The key equation for conversion is simple yet powerful: pH + pOH = 14.00 at 25°C. Therefore, knowing the pH of a solution allows for direct calculation of its pOH, and vice versa. This conversion is crucial when analyzing titration curves and determining the equivalence point.
Accurate pH measurements, often obtained using a pH meter, provide the necessary data for pOH calculation. This interconversion facilitates a comprehensive understanding of the solution’s acid-base properties throughout the titration process, aiding in precise concentration determination.
Utilizing KA and KB Values in Calculations
Acid and base dissociation constants, Ka and Kb, are vital for quantifying the strength of acids and bases, respectively, and are essential in titration calculations. Ka represents the extent of acid dissociation, while Kb indicates base dissociation.
These values are used to determine the pH of weak acid or base solutions and to calculate the hydrogen ion (H+) or hydroxide ion (OH–) concentrations. Furthermore, Ka and Kb are related by the ion product of water (Kw), allowing conversion between them (Ka * Kb = Kw).
During titrations, knowing these constants helps predict the titration curve’s shape and accurately determine the equivalence point. Utilizing these values ensures precise calculations of unknown concentrations, enhancing the reliability of analytical results.

Safety Considerations
Always wear appropriate PPE – gloves, goggles, and lab coats – when handling acids and bases. Proper chemical waste disposal is crucial for safety and environmental protection.
Handling Acids and Bases Safely
Dilute acids and bases, while seemingly less hazardous, still demand careful handling. Always add acid to water, never water to acid, to avoid violent exothermic reactions and potential splattering. Wear chemical-resistant gloves and safety goggles to protect skin and eyes from corrosive substances.
Work in a well-ventilated area, preferably under a fume hood, to minimize inhalation of fumes. Be aware of the specific hazards associated with each acid and base – consult Safety Data Sheets (SDS) for detailed information. Immediately neutralize any spills with appropriate neutralizing agents and clean up thoroughly.
Avoid contact with clothing and shoes; ensure proper storage away from incompatible materials. Remember, proactive safety measures are paramount when working with corrosive chemicals.
Proper Disposal of Chemical Waste
Neutralization is often a crucial first step in preparing acid or base waste for disposal, reducing its corrosivity. However, never mix incompatible chemicals – this can create dangerous reactions. Segregate waste streams into appropriately labeled containers, clearly identifying the contents.
Follow all local, state, and federal regulations regarding chemical waste disposal. Consult your institution’s environmental health and safety department for specific guidelines. Avoid pouring chemicals down the drain unless explicitly permitted and neutralized to a safe pH.
Utilize designated waste collection services for proper handling and treatment. Ensure containers are tightly sealed to prevent leaks or spills during transport. Responsible waste disposal protects both the environment and human health.
Personal Protective Equipment (PPE)
Safety goggles are paramount, shielding eyes from splashes or fumes during acid handling and titration procedures. Wear chemically resistant gloves – nitrile or neoprene are often suitable – to protect skin from corrosive substances. A lab coat provides a barrier against spills, safeguarding clothing and skin.
Closed-toe shoes are mandatory in the laboratory, preventing foot injuries from dropped glassware or chemical spills. Consider a face shield for procedures involving concentrated acids or potential splashing. Ensure PPE is in good condition, free from tears or punctures.
Proper PPE minimizes the risk of chemical exposure, promoting a safe working environment. Always remove PPE before leaving the lab to prevent contamination spread. Training on correct PPE usage is essential for all personnel.